The Main Cooling Cycle
A substance changes its state when the inherent amount of heat and varied. Ice is water in the solid state and steam vapor state water. Continuous changes liquid and liquid-vapor by the application of heat. Heat must be added or vaporize the boiling point of a substance. It should be collected for the dilution or strengthen substances. The amount of heat needed will depend on the content and change of pressure in the matter. Consider, for example, open a pot of boiling water heated by a gas flame. The boiling point of water at sea level 212Вєf (100C). Increase flame temperature and the water will boil from more rapidly, although the temperature does not change. Heat or boiling substances, heat must be removed from another substance. In this case, the heat is removed from the gas flame. Increase the temperature of the flame only the rate of heat transfer. It does not increase the temperature of the water.
The change in pressure will affect the boiling point of a substance. With increasing height above the sea level, atmospheric pressure and evaporating temperature drop.
For example, water will boil at 193F (89.4C) at an altitude of 10,000 feet. At pressures below 100 psig, the water has a temperature of boiling 338F (170C). The relationship pressure refrigeration this is shown in the following example. The tank contains a substance which evaporates at atmospheric pressure. However, it condenses to a liquid at 100 pounds of pressure. Liquid is from the tank through a hose and nozzle in a long coil of pipe to the atmosphere (see Fig. 4-10). 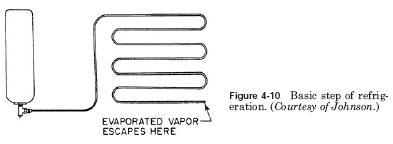
As the liquid enters the nozzle, his pressure is reduced to that of the atmosphere. This reduces the evaporation or boiling point. Part of the liquid evaporates or boils, using its own heat. In unevaporated liquid immediately cooled, his heat is taken away. The remaining liquid absorbs heat from rolled metal or tank and evaporates, cooling coil. Coil takes the heat from the surrounding space, cooling space. This unit will continue to provide cooling or cooling until the substance remains under pressure in the tank. All other components of the cooling system are only to enhance the cooling medium after it has done its job of cooling. Other parts of the cooling system in order of assemblies, tank or liquid receiver, expansion valve, evaporator, compressor, and capacitor.
Fig. 4-11 illustrates a typical system cooling cycle. The refrigerant is in the tank or liquid receiver under high pressure in the liquid state. When refrigerant enters the expansion valve, the pressure is lowered, and the liquid begins to evaporate. Complete evaporation occurs when the refrigerant passes into the evaporator. Evaporation, heat must be added to the refrigerant. In this case, the heat comes from the evaporator. As heat is removed from the coil, the coil cooling. The refrigerant is now under steam low pressure. Evaporator section of the system is often referred to low pressure, back pressure or suction side. Warm coil, the faster evaporation takes place and the higher suction pressure becomes. 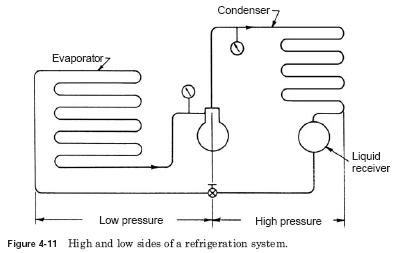
Compressor then takes the low vapour pressure and increases the pressure is enough for condensation of the refrigerant. This starts the high side of the system. To return a refrigerant in a liquid state (squeeze), the heat took the evaporator, compressor must be removed. This function of the condenser is used with air or water cooling coils. Be better than refrigerant, air or water absorbs heat. As it cools, the refrigerant is condensed into a liquid and comes in a liquid receiver or tank. As the refrigerant pressure was increased, it will condense at lower temperatures. In some systems, the liquid receiver may be part of another device, such as evaporator or condenser. ..
|

